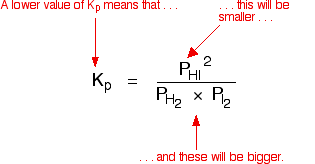
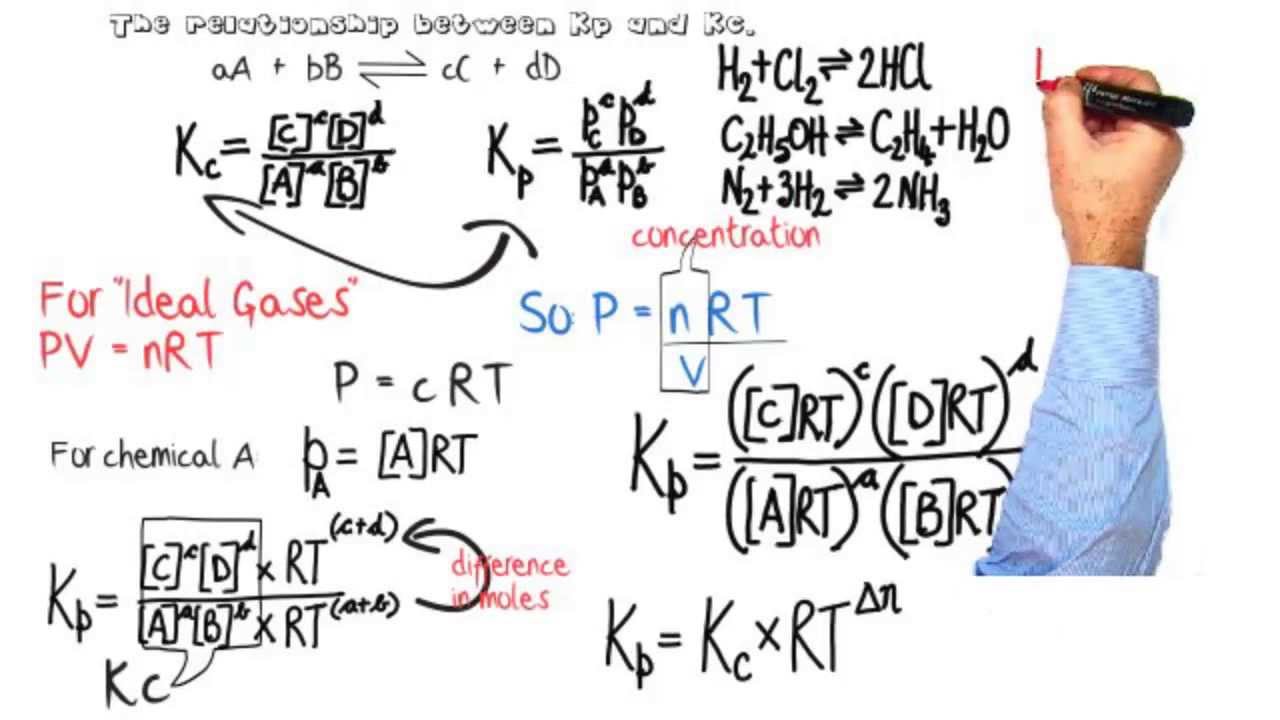
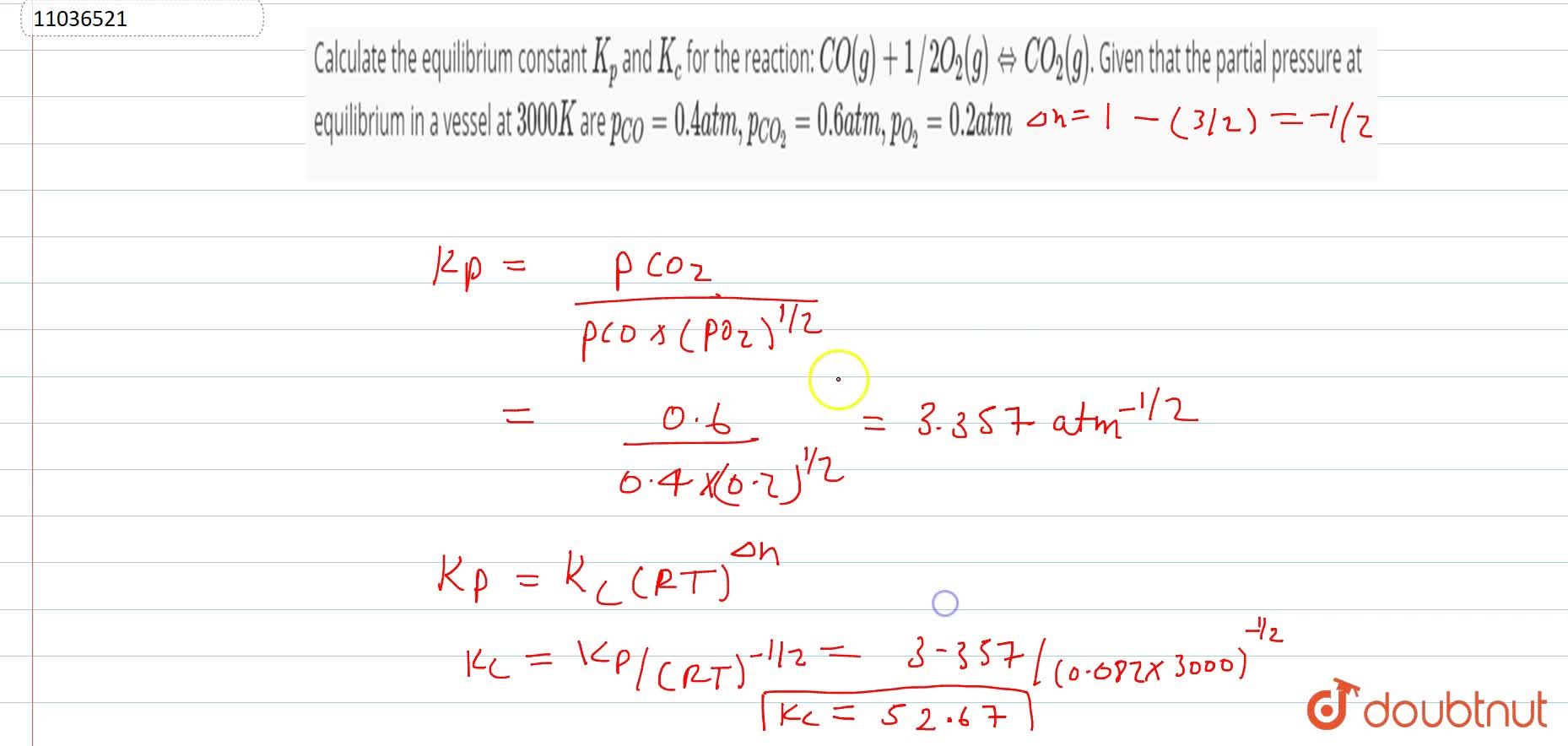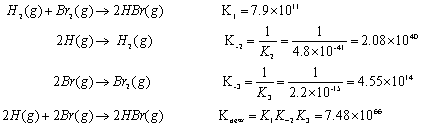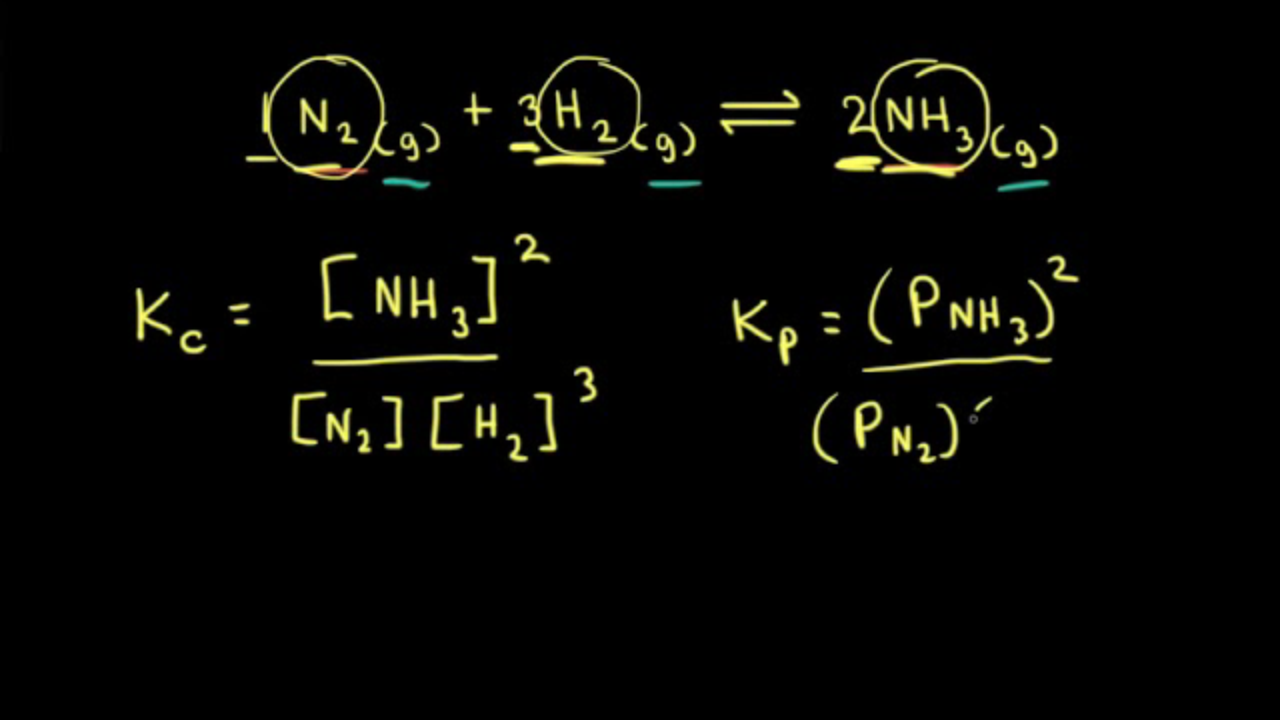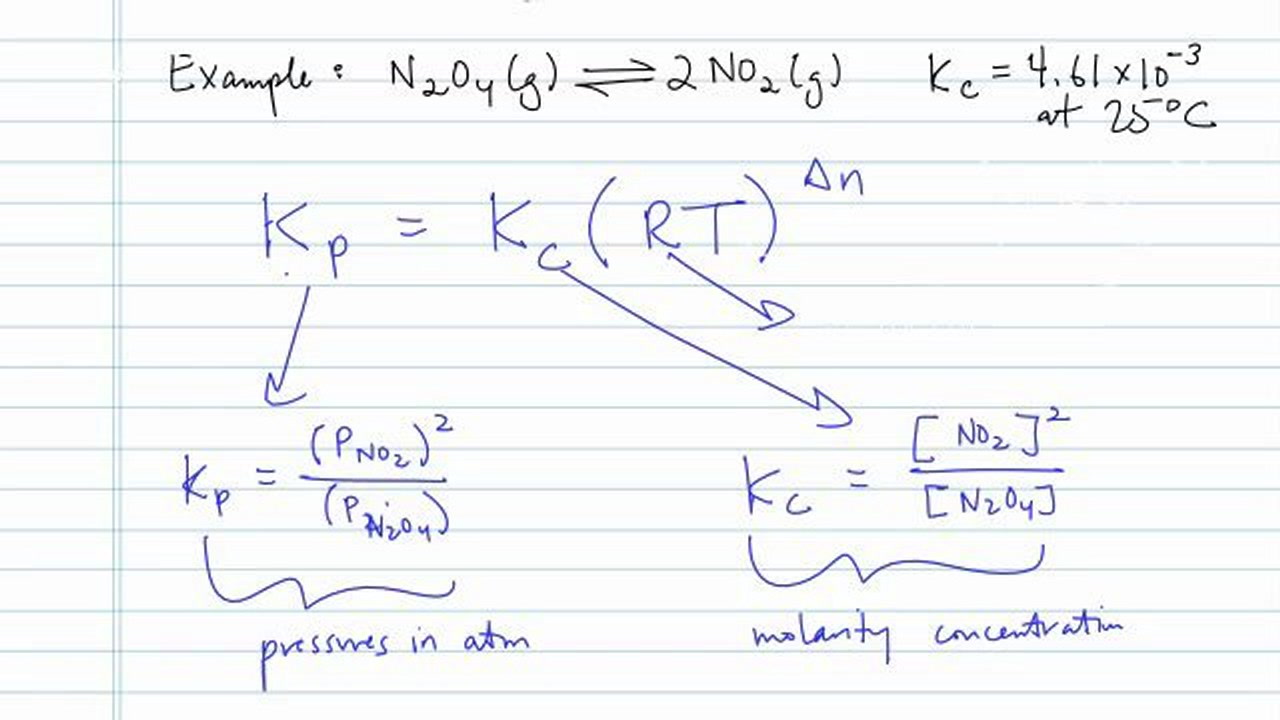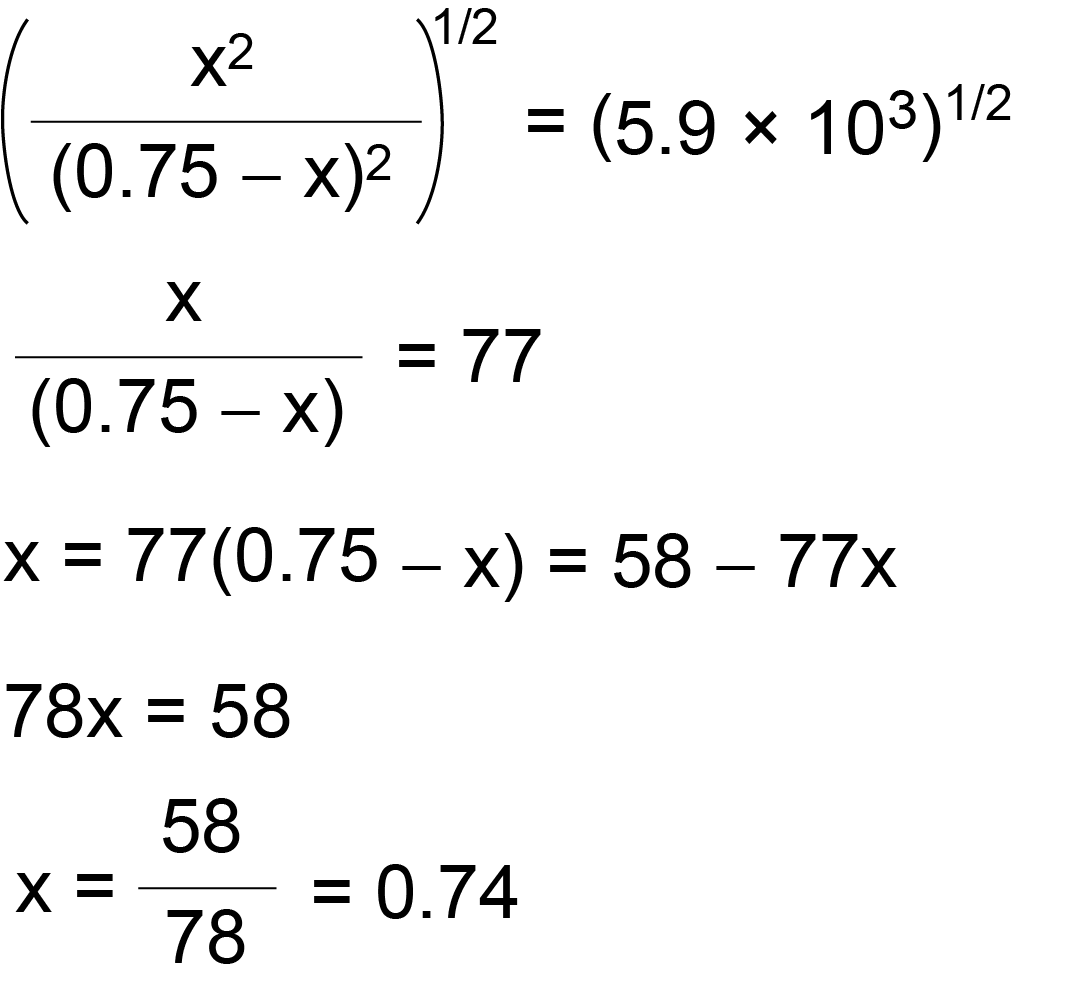Learn how the equilibrium constants Kc and Kp are related. | Chemistry help, Ap chemistry, High school chemistry

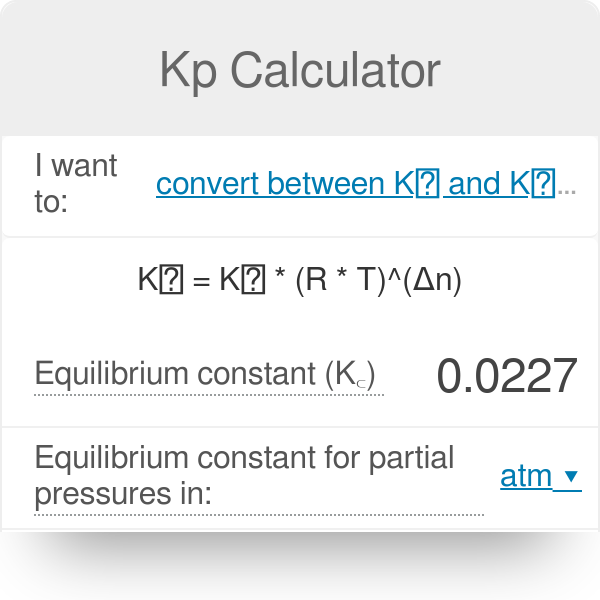
Learn how to calculate an equilibrium constant Kp. | Chemistry help, Teaching chemistry, High school chemistry

Chapter 15. Overview Equilibrium Reactions Equilibrium Constants K c & K p Equilibrium Expression product/reactant Reaction Quotient Q Calculations Le. - ppt download